 This calculation is fundamental in chemistry for various purposes, including determining the amount of reactant or product in a chemical reaction and understanding the composition of chemical compounds. To use the calculator, you input the mass of the substance and its molar mass, and it will provide you with the moles per gram. “Molar Mass of Substance” is the molar mass of the substance in grams per mole (g/mol). 
2: Conversion from number of particles to mass, or from mass to number of particles requires two steps. This chemistry video tutorial explains the conversion process of atoms to grams which is a typical step in common dimensional analysis stoichiometry problems. In order to convert from mass to number of particles or vice-versa, a conversion to moles is required. Mass and number of particles are both related to grams. Molarity is measured in moles per liter (mol/L). We can combine the two types of problems into one. Is molarity in grams per mole No, molarity is not in grams per mole. Use the formula: grams (molality molar mass of solute mass of solvent) / 1000. “Mass of Substance” is the mass of the substance in grams. To convert molality to grams, you need to know the mass of the solvent and the molality of the solute.ApexBio by An Apoptosis and Epigenetics Company. The answer of 19.713 mg appears in the Mass box. Enter 10 into the Concentration box and select the correct unit (millimolar) Enter 10 into the Volume box and select the correct unit (milliliter). This means that one mole of carbon atoms weighs approximately 12.01 grams. Enter 197.13 into the Molecular Weight (MW) box. How many grams in a mole: The molar mass of carbon (C) is approximately 12.01 g/mol. “Moles Per Gram” is the moles of the substance per gram. The grams to moles calculator bridges the gap between these two units, allowing chemists to translate mass into the corresponding number of particles.Moles Per Gram (moles/g) = Mass of Substance (grams) / Molar Mass of Substance (g/mol) The formula for calculating moles per gram is as follows: This calculation is essential for various chemical applications, especially in stoichiometry and determining the molar mass of compounds. Whether you’re a student or a professional chemist, this tool will help you in various chemistry-related tasks.About Moles Per Gram Calculator (Formula)Ī Moles Per Gram Calculator is a useful tool in chemistry that helps determine the number of moles of a substance per gram of that substance. Molar mass of NaCl is 58.443, how many grams is 5 mole NaCl grams 58.443 × 5 292.215 (g) Molar mass of AgNO3 is 169. With the G/Mol to Grams Calculator and the provided formula, you can easily calculate missing values and make your chemical calculations more efficient. The mass (in grams) of a compound is equal to its molarity (in moles) multiply its molar mass: grams mole × molar mass. Understanding the conversion between grams per mole, grams, and moles is essential in chemistry. Just ensure you have accurate values for the given variables. Q3: Can I use the calculator for both elements and compounds?Ī3: Yes, the calculator works for both elements and compounds. This value is often listed in periodic tables for elements. Q2: How do I find the grams per mole value of a substance?Ī2: To find the g/mol value, divide the mass of a substance in grams by the number of moles. FAQs Q1: Why is it important to convert between g/mol, grams, and moles in chemistry?Ī1: Converting between these units is crucial for various chemical calculations, such as determining reaction stoichiometry and finding the mass of a substance in chemical reactions. So, 36 grams of the substance are present in 3 moles. Note that under the conditions stated in the problem, one mole of any gas, considered ideal, occupies a volume of approximately 22. 2) Now, convert the volume of CO 2 into the amount of substance (moles). 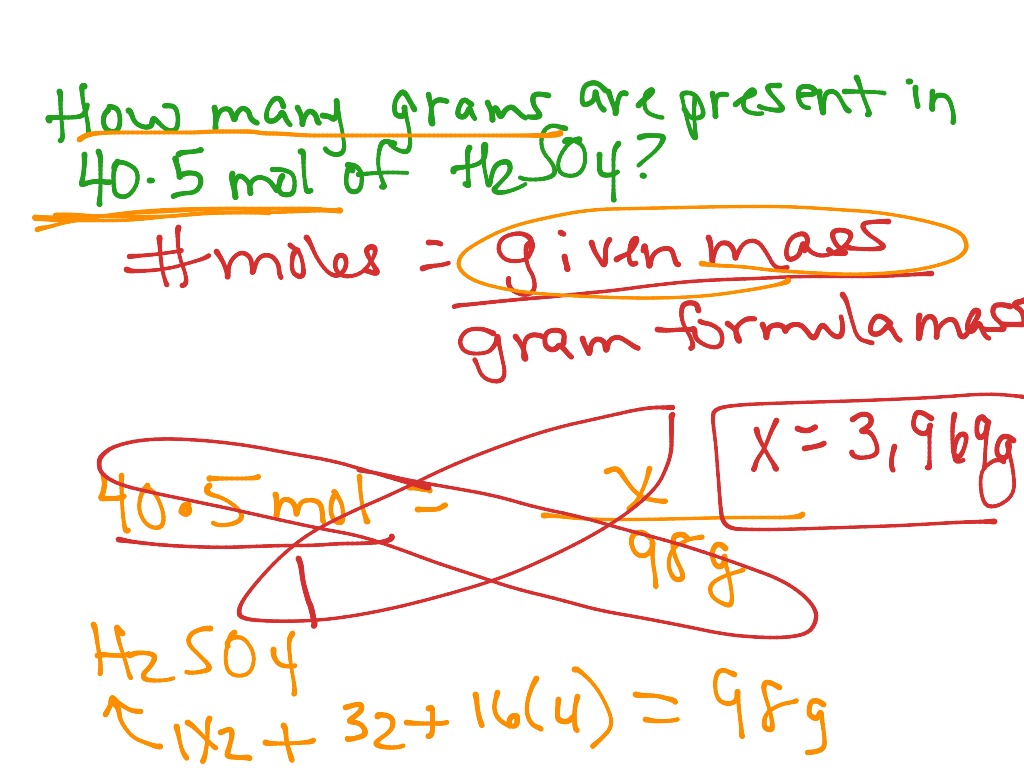
The calculator will instantly calculate the results using the chemical equation for moles. Let’s illustrate how to use the formula with an example:Įxample: You have 12 g/mol of a chemical substance, and you want to find out how many grams are present in 3 moles. The ‘Chemical Equation’ field in the calculator is now highlighted in light pink, indicating that the equation we entered was unbalanced. Mole Calculator Select the chemical parameter (mass, moles, molecular weight) from the list and provide it in required ones. The formula for converting between grams (G), moles (mol), and grams per mole (g/mol) is: 
It’s a quick and efficient way to solve chemistry problems. Simply enter two of the three variables: grams, moles, or grams per mole, and let the calculator determine the missing value for you. Using the G/Mol to Grams Calculator is a breeze. Whether you need to find the weight of a chemical substance in grams or calculate the number of moles from its g/mol value, this guide will help you understand the process and provide a convenient G/Mol to Grams Calculator to simplify your calculations. Converting between grams per mole (g/mol), grams, and the number of moles is a fundamental concept in chemistry. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
